|
This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. 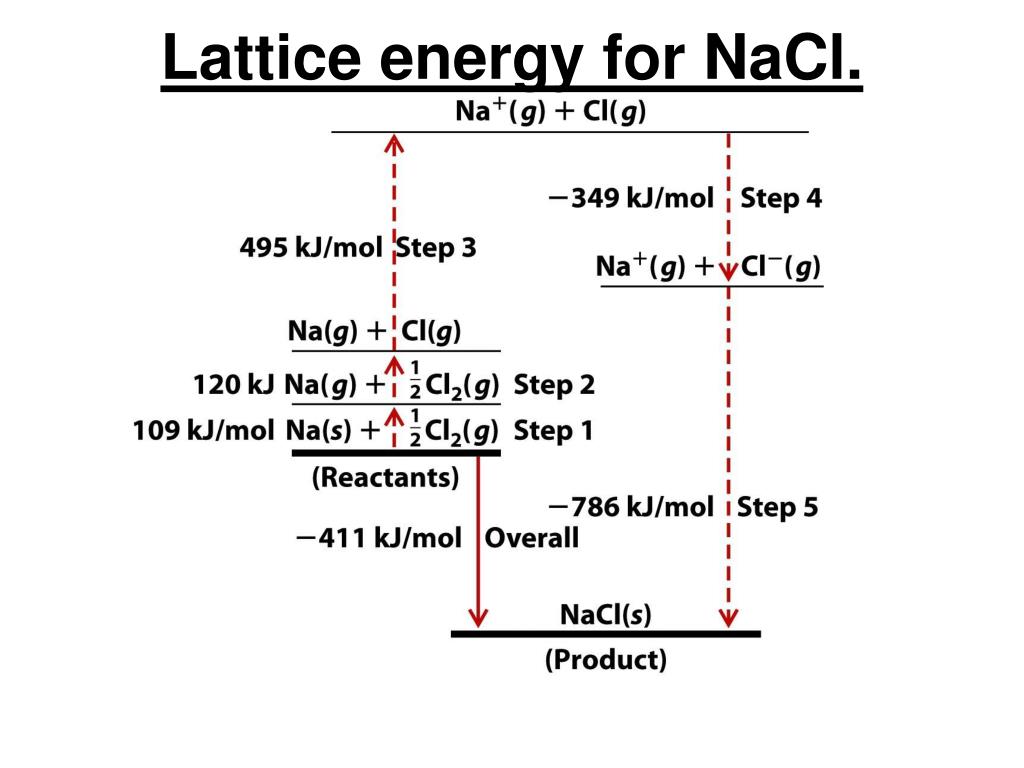
first ionization energy of K = 418.7 kJ/mol. The greater the lattice enthalpy, the stronger the forces. Lattice enthalpy is a measure of the strength of the forces between the ions in an ionic solid. The lattice formation enthalpy is the enthalpy change when 1 mole of solid crystal is formed from its separated gaseous ions. Lattice dissociation enthalpies are always positive. The lattice dissociation enthalpy is the enthalpy change needed to convert 1 mole of solid crystal into its scattered gaseous ions. What is the lattice enthalpy of dissociation? Energy is required to break apart the solid, so the lattice energy will always be positive. Lattice energy is the amount of energy required to seperate the crystals into gaseous ions. How is lattice energy related to solubility?Īs lattice energy increases, solubility will decrease. It is the energy required to entirely separate one mole of ionic compound into gaseous ions. What is Lattice Enthalpy? In simple words, Lattice enthalpy is the change in Enthalpy connected with the formation of one mole or an ionic compound from its gaseous ions, other things remaining standard. Alternately, it can be defined as the energy that must be supplied to one mole of an ionic crystal in order to separate it into gaseous ions in a vacuum via an endothermic process. Lattice energy can be defined as the energy required to convert one mole of an ionic solid into gaseous ionic constituents. Formula and structure: CaCl2 is the chemical calcium chloride formula and its molar mass is 110,983 g/mol. Calcium chloride is an essential salt of calcium that has many applications for household and industrial use. The lattice energy of CaCl2 is –2195.2 kJ/mol. Rubidium iodide has a lattice energy of -617 kJ/mol, while potassium bromide has a lattice energy of -671 kJ/mol. Which iodide has the highest lattice energy? Conversely, for a given alkali metal ion, the fluoride salt always has the highest lattice energy and the iodide salt the lowest. Which has highest lattice energy?īecause the ionic radii of the cations decrease in the order K + > Na + > Li + for a given halide ion, the lattice energy decreases smoothly from Li + to K +. KCl will have the more negative lattice energy because potassium is smaller than cesium and they both have a 1 + charge. Which has higher lattice energy LiCl or KCl?Įxplain your choices. 
The energy value can be estimated using the Born-Haber cycle, or it can be calculated theoretically with an electrostatic examination of the crystal structure. Lattice energy cannot be determined experimentally due to the difficulty in isolating gaseous ions. The lattice energy for potassium iodide, KI, is 627.2 kJ/mol. What is the lattice energy of potassium iodide? What is the crystal lattice energy of KI? Lattice formation energy: 6 What is the lattice enthalpy of dissociation?.1 What is the crystal lattice energy of KI?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
